50fc2ee2...
JEE Physics
Why Doubling Light Intensity Doesn't Double Electron Energy
Go beyond formulas. Understand why Einstein's photoelectric effect revolutionized physics - and how it appears in JEE.
Click to zoom
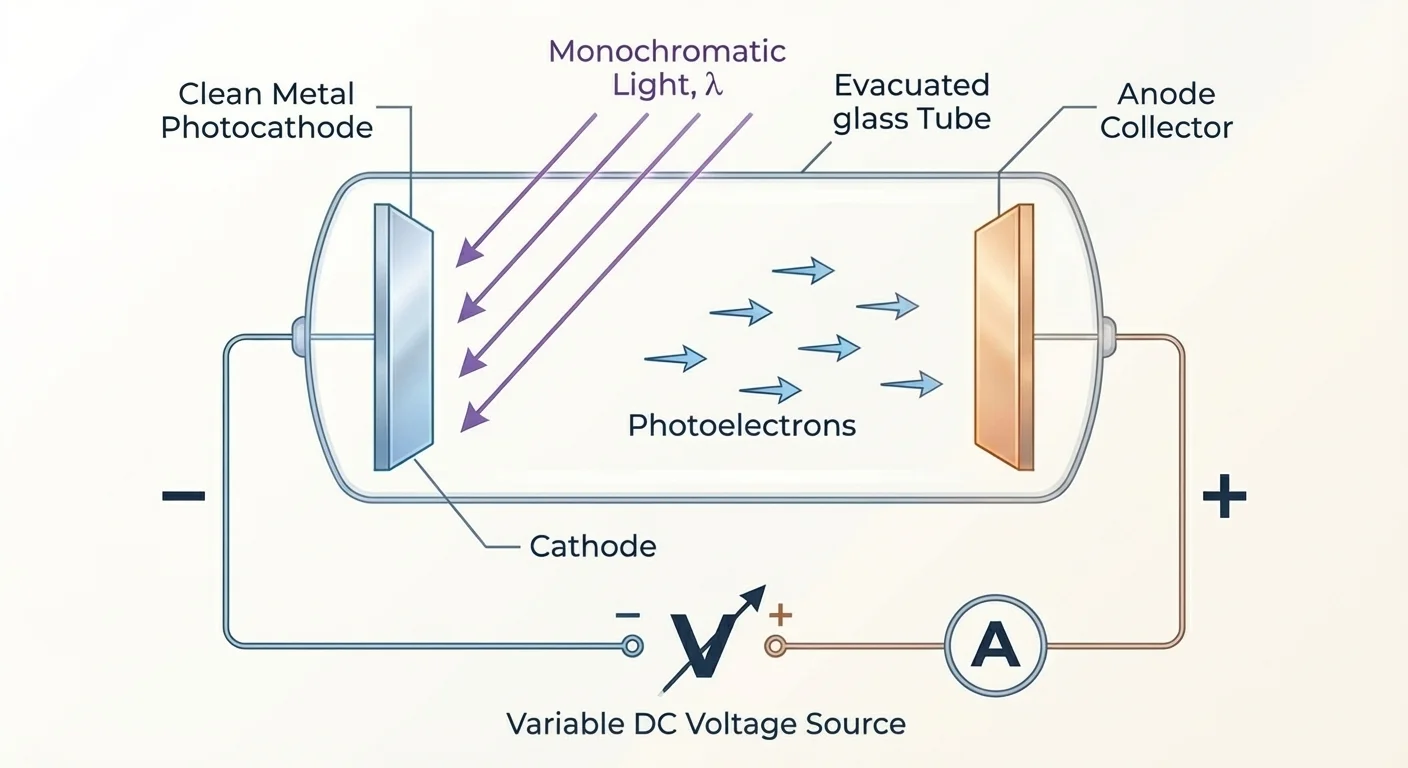
Experimental setup for studying the photoelectric effect
When electromagnetic radiation of sufficiently high frequency strikes a metal surface, electrons are emitted from the surface. This phenomenon is called the photoelectric effect.
Key equation: Maximum kinetic energy of photoelectron
where is Planck's constant , is the frequency of incident light, and is the work function of the metal.
1 / 4
If the intensity of incident light is doubled while keeping the frequency constant (above threshold), what happens to the maximum kinetic energy of emitted photoelectrons?